Home »Sourcing Solutions » Pregabalin
What is Pregabalin
Pregabalin is a 3-isobutyl derivative of gamma-aminobutyric acid (GABA) with anti-convulsant, anti-epileptic, anxiolytic, and analgesic activities. It was approved for medical use in the United States in 2004 and was developed as a successor to gabapentin. Pregabalin is classified as a Schedule V controlled substance in the United States under the Controlled Substances Act of 1970. In the United Kingdom, it is classified as a Class C controlled substance.
It is a gabapentinoid and acts by inhibiting certain calcium channels. Pregabalin comes as a capsule, a solution, and an extended-release tablet. All forms are taken by mouth. It is generally used to treat epilepsy, neuropathic pain, fibromyalgia, restless leg syndrome, opioid withdrawal and generalized anxiety disorder.
| PRODUCT SPECIFICATIONS | |
|---|---|
| Name of Product | Pregabalin |
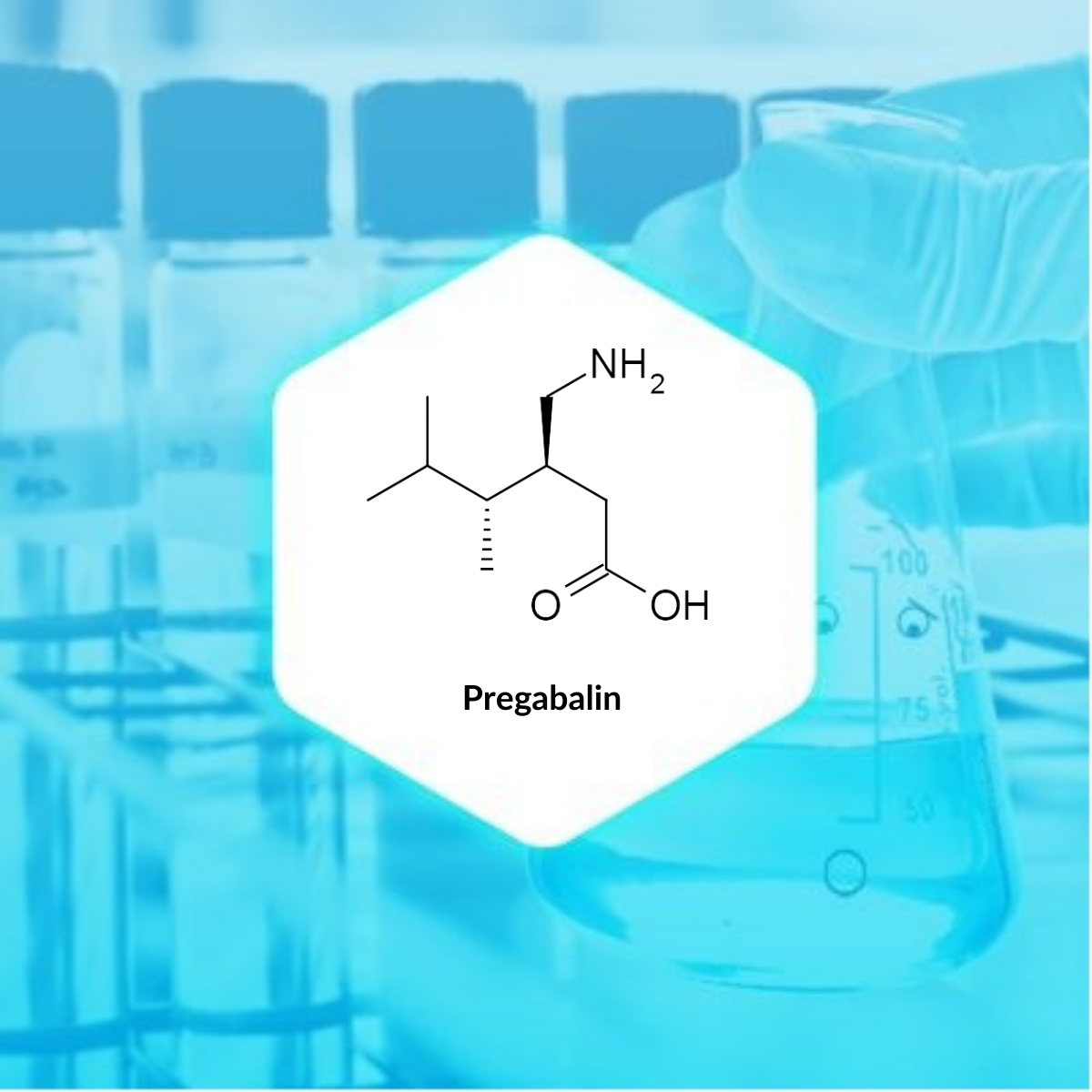
| IUPAC Name | (3S)-3-(aminomethyl)-5-methylhexanoic acid |
| Synonyms | Pregabalin, Lyrica, 3-isobutyl GABA, (S)-3-(Aminomethyl)-5-methylhexanoic acid |
| CAS No | 148553-50-8 |
| Molecular Formula | C8H17NO2 |
| Molecular Weight | 159.23 g/mol |
| Pubchem CID | 5486971 |
| Pubchem SID | 463751341 |
Chemical Properties
| SR. No | Criteria | Limit/Specification |
|---|---|---|
| 1 | Appearance (Color) | White to off-white |
| 2 | Appearance (Form) | Crystalline Solid |
| 3 | Solubility | Freely soluble in water |
| 4 | Melting Point | 176 - 178ºC |
| 5 | Boiling Point | 144 - 147 ºC | 6 | Uses | Seizures, Neuropathic Pain, Anxiety disorders |
Uses
- Pregabalin is used in combination with other medications to treat Seizures.
- It is the first-line agent for the treatment of pain associated with diabetic neuropathy, post-herpetic neuralgia, central neuropathic pain, and fibromyalgia.
- Pregabalin is a fairly effective and safe medication for treating generalised anxiety disorder. It appears to have anxiolytic effects similar to benzodiazepines with less risk of dependence.
- There is a minimal benefit and high risk in patients with chronic low back pain, according to the evidence. As of 2016, there is limited evidence of benefits in alcohol withdrawal and detox from several other drugs.
Side effects
The more common side effects of pregabalin include:
- Dizziness
- Sleepiness
- Trouble concentrating
- Blurry vision
- Dry mouth
- Weight gain
- Swelling of your hands or feet
Serious side effects include:
- Serious allergic reactions
- Suicidal thoughts or actions
- Heart problems
- Serious breathing problems
Addiction may happen when pregabalin is taken in high dosages over a long period of time, but the risk is low when taken in normal amounts. The safety of using it during pregnancy or breastfeeding is unknown.
FAQs
Q. Is Pregabalin a controlled substance?
Pregabalin is, indeed, a regulated substance. It is a prescription medicine classed under schedule V. Schedule V medicines have established medical usefulness, but they can also develop psychological or physical dependence. This suggests that the substance could be abused.
Q. Is Pregabalin the same as Lyrica?
Lyrica is a brand name prescription medication containing Pregabalin as the active pharmaceutical ingredient.
Q. How Pregabalin differs from Gabapentin?
- Pregabalin has an increased binding affinity for the alpha-2-delta protein and is a more potent analgesic in neuropathic pain compared with gabapentin.
- Pregabalin is more likely than gabapentin to cause side effects such as dry mouth, constipation, swelling (oedema), or weight gain.
- Pregabalin is absorbed faster and starts working more quickly than gabapentin.
- Pregabalin may have a higher addiction potential than gabapentin due to its faster absorption and onset of action.
