Gemifloxacin mesylate is the mesylate salt of gemifloxacin, which is a naphthyridine and fluoroquinolone derivative, antibacterial agent. It is a quinolone with a broad-spectrum activity used in the treatment of acute bacterial exacerbation of chronic bronchitis and mild-to-moderate pneumonia. It is available in oral formulations, commonly sold under the trade name Factive.
Table of Contents
Gemifloxacin Mesylate Export Insights
Gemifloxacin Mesylate API market
Major exporting countries
India is the largest exporter of API. China is another supplier for the API other than India but is exporting smaller quantities.
Export from India
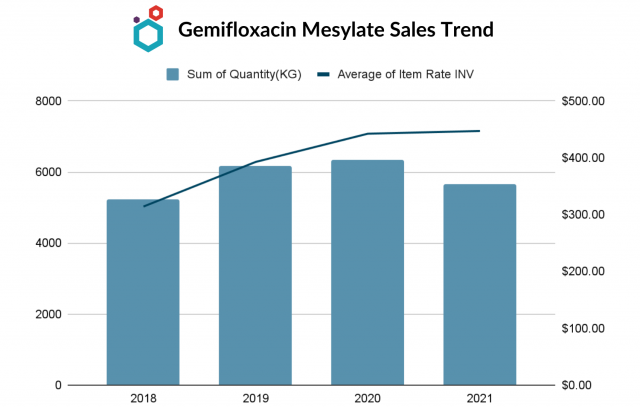
- The export of the API has been increasing and has fallen in 2021.
- The average rate/kg has been showing an uptrend with the increase in quantity and has steadied in 2021.
– The Top companies are :
- Maithri Drugs
- Honour Labs
- Hetero Drugs
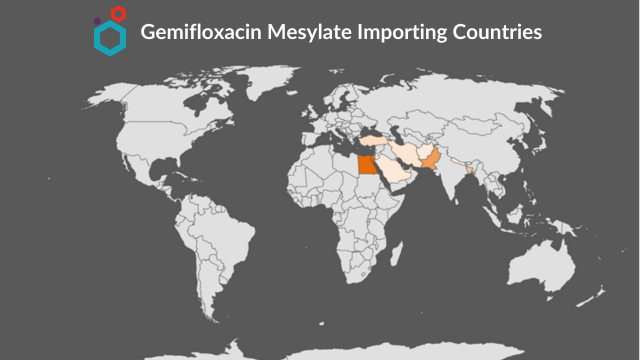
Major importing countries
- Egypt, Bangladesh, and Pakistan have been the larger importers of APIs from India.
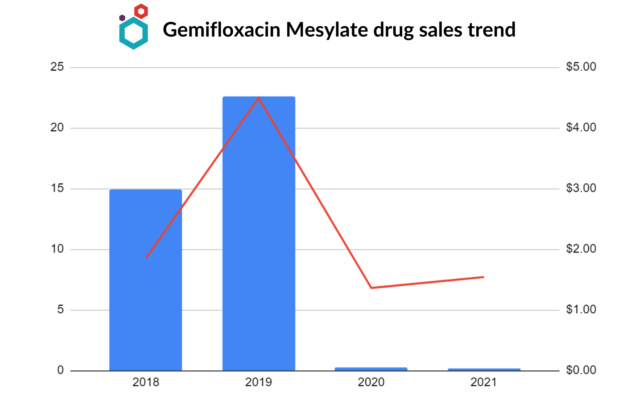
Gemifloxacin Mesylate Drug Market
Major Importing Countries
Nigeria and Myanmar are the two key markets importing from India.
Other countries are:
- Malaysia
- Palau
- Singapore
This is just a snapshot of the research that we have done on Gemifloxacin Mesylate. This is all that the full research report covers:-
- Competitive landscape comprising key countries, and companies.
- Comprehensive Coverage of US and other key markets and other crucial information.
- Complete coverage of both Qualitative and Quantitative Inputs about the drug.
For a negligible price you can buy the full report from us.
For the entire research report on Gemifloxacin Mesylate, send us an enquiry-
Macsen Labs is the supplier of high-quality Gemifloxacin Mesylate.
Disclaimer–
The information provided here is based on general knowledge, articles, research publications etc. and we do not claim the authenticity of any of the information provided above. We do not claim or suggest/advise any medical, therapeutic, health or nutritional benefits of Gemifloxacin Mesylate. We do not supply or promote our Gemifloxacin Mesylate product for the applications which are covered by valid patents and which are not approved by the FDA.
