Prilocaine is a toluidine derivative and is a secondary amide analogue of lidocaine. It is used as a general local anaesthetic drug for pain relief with a prolonged duration of action and rapid onset than other similar medicines.
Prilocaine Export Insights
Prilocaine API market
Exporting countries
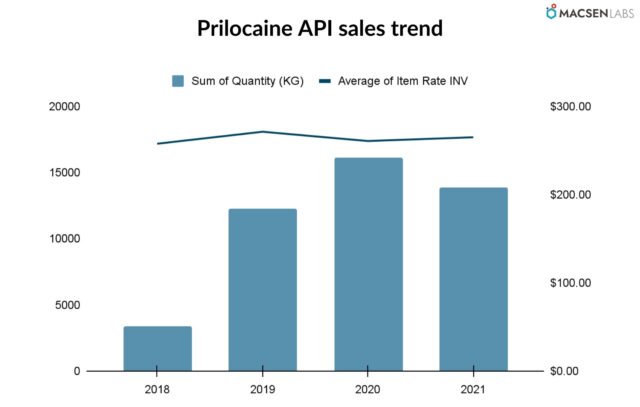
Export from India
- The annual quantity has been increasing year on year till 2020, but it fell in 2021.
- The average price has seen a downward trend after going upwards. and has been steady since.
The three companies exporting are:
- Gufic Lifesciences
- HRV global Sciences
- Swati Spentose Pvt. LTD.
Export from other countries
Apart from India, Belgium and China have also shipped API as per the data available in the public domain.
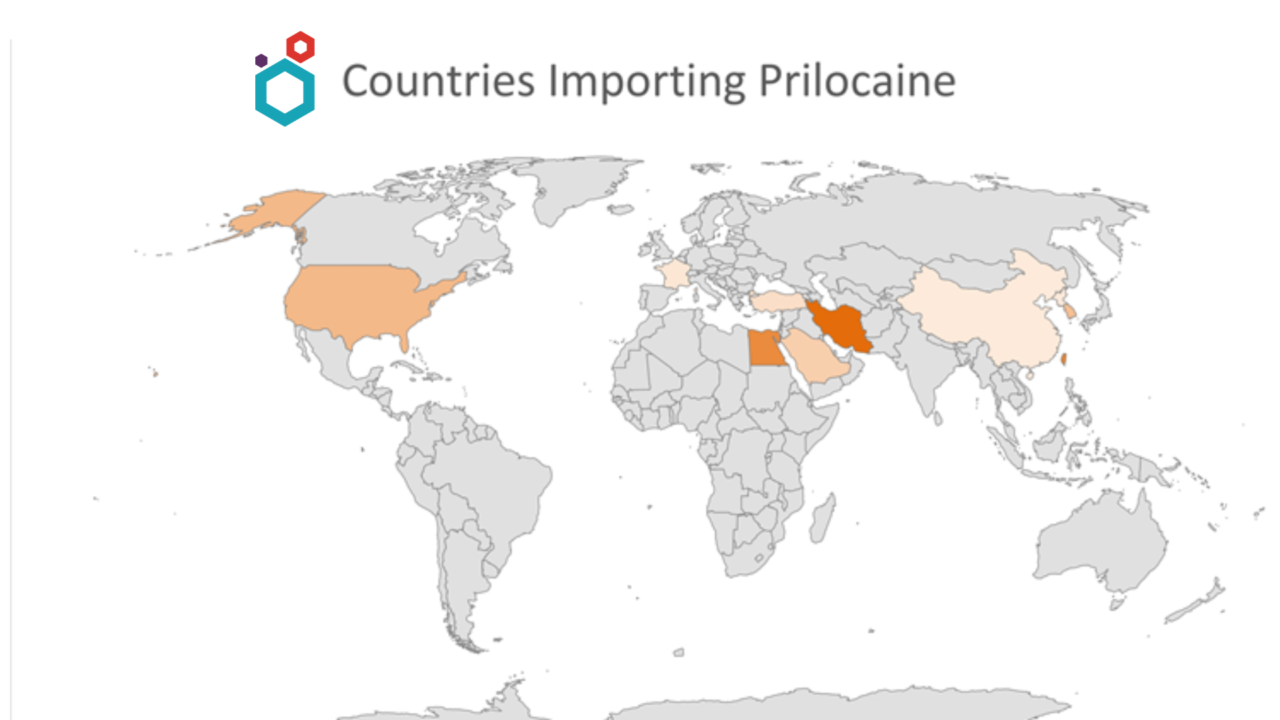
Major importing countries
- Major Countries importing are Taiwan and Egypt.
Other Major Countries are:
- Turkey
- USA
- South Korea
This is just a snapshot of the research that we have done on Atovaquone. This is all that the full research report covers:-
- Competitive landscape comprising key countries, and companies.
- Comprehensive Coverage of US and other key markets and other crucial information.
- Complete coverage of both Qualitative and Quantitative Inputs about the drug.
For a negligible price you can buy the full report from us. Drop us-
For the entire research report on Prilocaine, send us an enquiry-
Macsen Labs is the supplier of high-quality Prilocaine.
