Home » Organic Fine Chemicals and Intermediates » 2-Aminopyrimidine
Many pharmacological and agricultural products use 2-aminopyrimidine as a key component. The pyrimidine ring is also found in life-sustaining chemicals including vitamins, coenzymes, and uric acid, as well as medications like Veronal, Sulfadiazine, Fluorouracil, Glivec, and Rosuvastatin. Because the aminopyrimidine ring is a portion of nucleotide bases in DNA and RNA, which are the most significant components of living organisms, their importance in nature cannot be overstated.
Because 2-aminopyrimidine derivatives have such a broad range of actions, the organic synthesis of these compounds has attracted interest for decades, and the development of new techniques for the synthesis of these compounds is of great interest.
One of the most significant pharmacophores responsible for the biological features of its derivatives is the heterocyclic 2-aminopyrimidine system.
| PRODUCT SPECIFICATIONS | |
|---|---|
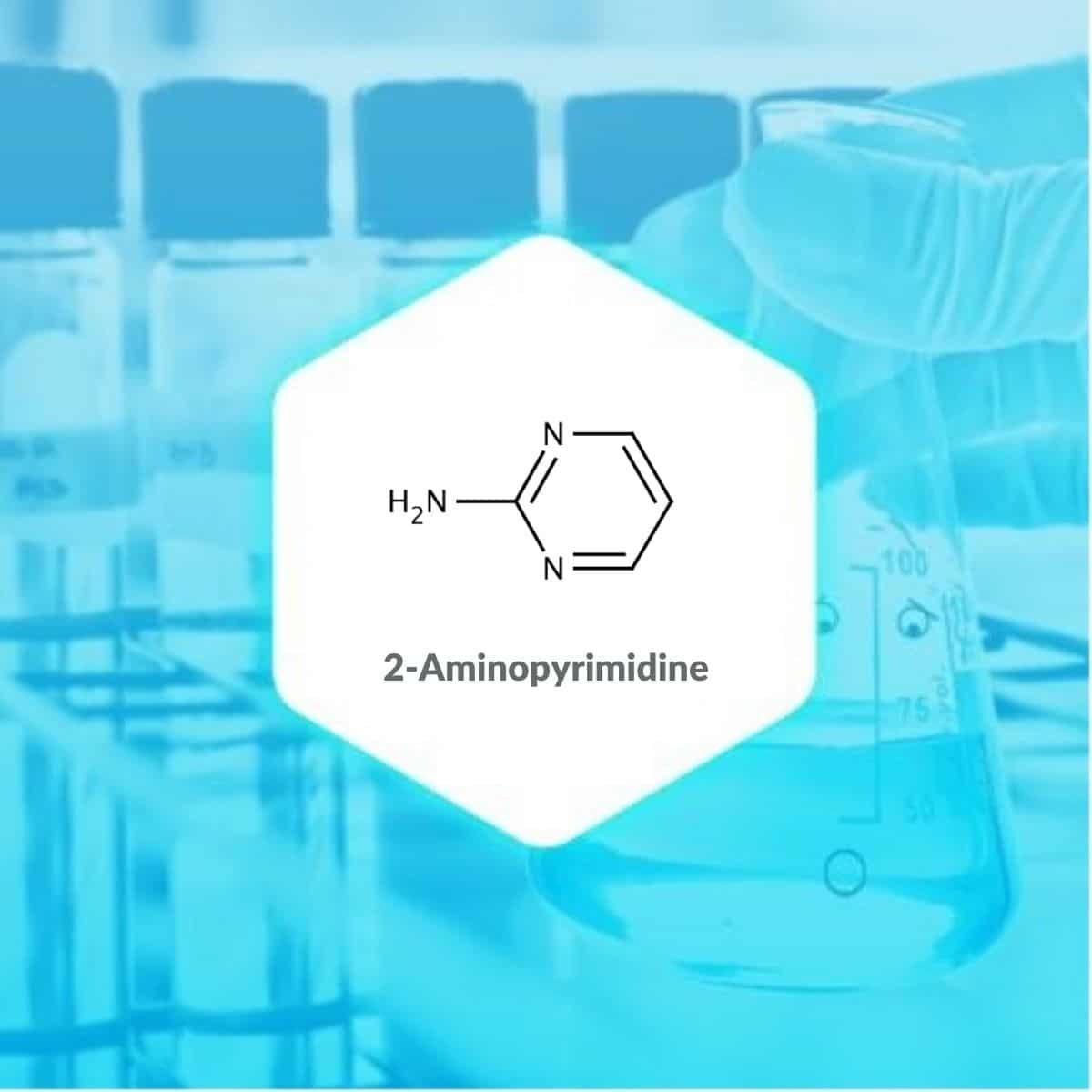
| Name of Product | 2-Aminopyrimidine |
| CAS NO | 109-12-6 |
| SYNONYMS | pyrimidin-2-amine; 2-Pyrimidinamine; Pyrimidinamine; 2-aminopirimidina |
| MOLECULAR FORMULA | C4H5N3 |
| MOLECULAR WEIGHT | 95.10 g/mol |
| USES | in the treatment of allergic conditions and gastric ulcers |
| Pubchem CID | 7978 |
| Pubchem SID | 462770343 |
TECHNICAL SPECIFICATIONS
| SR. No | Criteria | Limit/Specification |
|---|---|---|
| 1 | Appearance (COLOR) | White to off white |
| 2 | APPEARANCE (FORM) | POWDER OR CRYSTALS |
| 3 | Infrared Spectrum | CONFORMS TO STRUCTURE |
| 4 | TITRATION (NT) HCLO4 0.1M | 96.5 - 103.5 % |
| 5 | PURITY (GC AREA %) | ≥ 96.5 % |
| 6 | SOLUBILITY | Soluble in water |
Buy high purity 2-Aminopyrimidine from the leading manufacturer with ISO certification. For buying, send us an enquiry-
