Home »Organic Fine Chemicals and Intermediates » Michler’s ketone
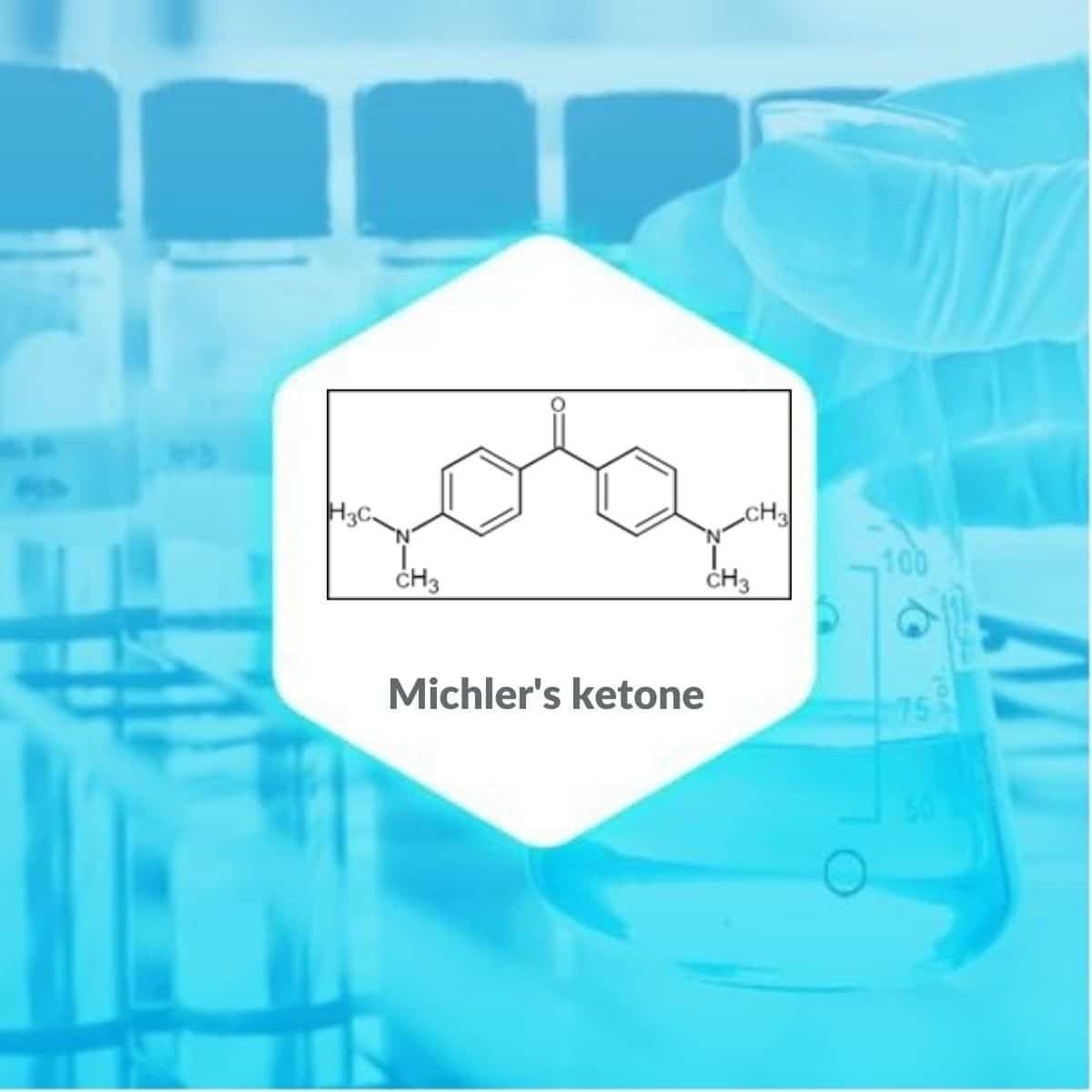
Michler’s ketone is an organic compound that appears as blue powder or white to greenish crystalline powder having the molecular formula [(CH3)2NC6H4]2CO. It is named after Wilhelm Michler, a German chemist. Several of the methyl violet dyes, such as crystal violet, are made by condensation of different aniline derivatives with Michler’s ketone.
Because of its absorption qualities, Michler’s ketone is a chemical intermediary in the production of dyes and pigments and is used in the dyeing of paper, textiles, and leather. It is also used as a photosensitizer. When heated to decomposition, Michler’s Ketone produces hazardous nitrogen oxide emissions. The central nervous system can be affected by Michler’s ketone.
| PRODUCT SPECIFICATIONS | |
|---|---|
| Name of Product | Michler's ketone |
| CAS NO | 90-94-8 |
| SYNONYMS | bis[4-(dimethylamino)phenyl]methanone |
| MOLECULAR FORMULA | C17H20N2O |
| MOLECULAR WEIGHT | 268.35 |
| MELTING POINT | 173 °C (343 °F; 446 K) |
| USES | to visualize ureteral orifices during cystoscopy and ureteral catheterization. |
| Pubchem CID | 7031 |
| Pubchem SID | 462770345 |
TECHNICAL SPECIFICATIONS
| SR. No | Criteria | Limit/Specification |
|---|---|---|
| 1 | Appearance (COLOR) | BLUE TO GREY |
| 2 | APPEARANCE (FORM) | POWDER |
| 3 | INFRARED SPECTRUM | CONFORMS TO STRUCTURE |
| 4 | CARBON CONTENT | 74.2 - 78.0 % |
| 5 | PURITY (HPLC AREA %) | ≥97.5 % |
| 6 | NITROGEN CONTENT | 10.0 - 10.8 % |
For buying, send us an enquiry-
