What is Rilpivirine?
Rilpivirine is an aminopyrimidine derivative, commonly sold under the brand names Edurant and Rekambys. It is a second-generation non-nucleoside reverse transcriptase inhibitor (NNRTI). It is a prescription drug approved by the U.S. Food and Drug Administration (FDA) for use with other HIV medicines to treat HIV infection. The drug comes in tablet form and as an intramuscular injection that is given once a month or every two months.
| PRODUCT SPECIFICATIONS | |
|---|---|
| Name of Product | Rilpivirine |
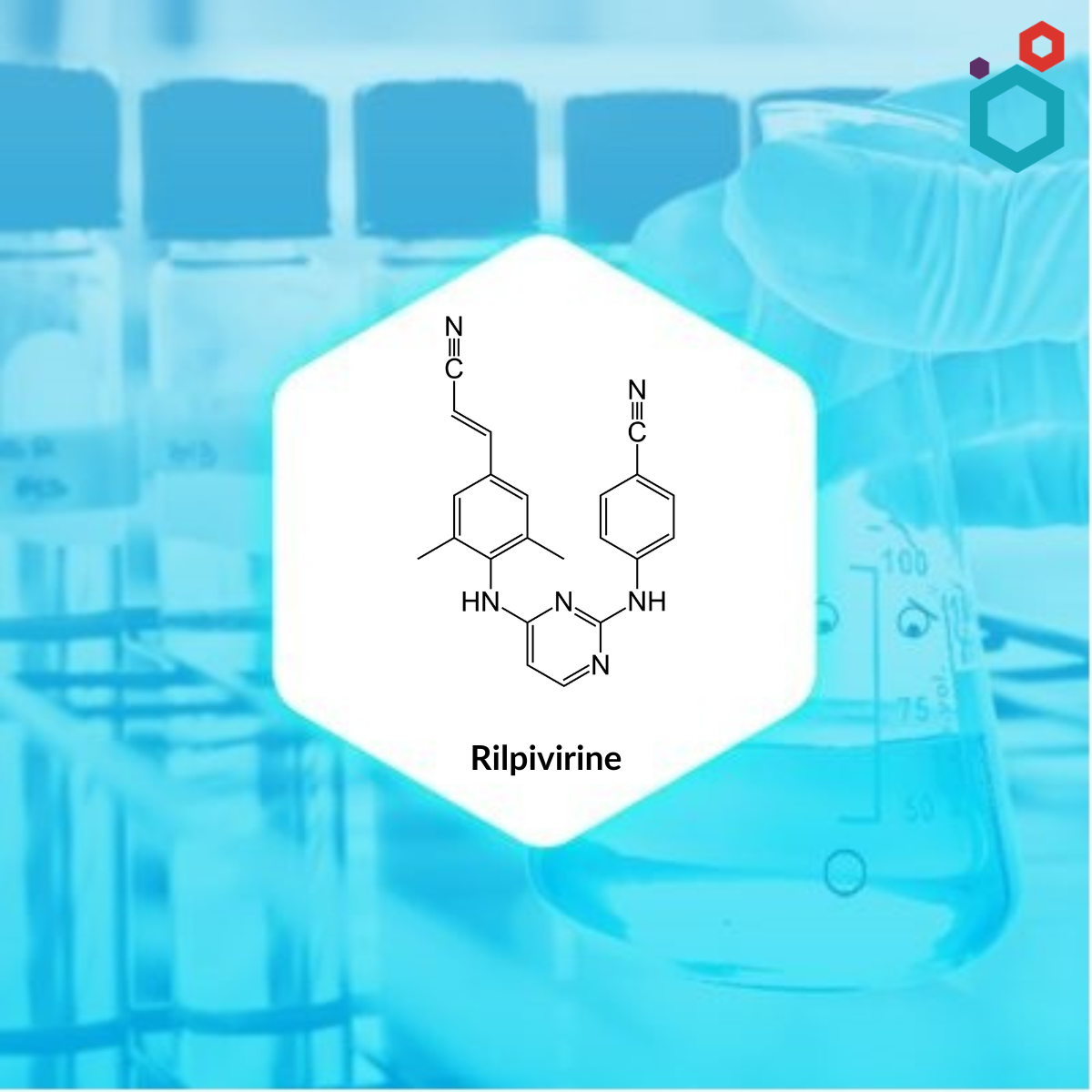
| IUPAC Name | 4-[[4-[4-[(E)-2-cyanoethenyl]-2,6-dimethylanilino]pyrimidin-2-yl]amino]benzonitrile |
| Synonyms | TMC278; Edurant; Rilpivirine free base; Rilpivirina; Rilpivirine |
| CAS No | 500287-72-9 |
| Molecular Formula | C22H18N6 |
| Molecular Weight | 366.4 g/mol |
| Pubchem CID | 6451164 |
| Pubchem SID | 470087776 |
Chemical Properties
| SR. No | Criteria | Limit/Specification |
|---|---|---|
| 1 | Appearance (Form) | Solid Powder |
| 2 | Appearance (Colour) | White to off-white, Slightly yellow |
| 3 | Melting Point | 241-243°C |
| 4 | Solubility | Readily soluble in DSMO (> 50 mg/mL) Moderately soluble in PEG 400 (40 mg/mL) Practically insoluble in water (20 mg/mL) |
| 5 | Other Experimental Property | Hygroscopic and light sensitive |
Mechanism of Action
Rilpivirine is a non-nucleoside reverse transcriptase inhibitor (NNRTI). When HIV infects a cell, an enzyme reverse transcriptase copies the viral single-stranded RNA genome into double-stranded viral DNA. The viral DNA is subsequently incorporated into the chromosomal DNA of the host, allowing the virus to replicate through cellular processes such as transcription and translation. RTIs block reverse transcriptase’s enzymatic function and prevent completion of synthesis of the double-stranded viral DNA, thus preventing HIV from multiplying.
Uses
- Rilpivirine is used in combination with other medications to treat human immunodeficiency virus type 1 (HIV-1) in specific adults and children, 12 years and older who weigh at least 77 lb (35 kg) and who have never had antiretroviral therapy.
- It is also used in combination with cabotegravir (Vocabria) to treat HIV-1 infection in adults for a limited time.
Side effects
The most common side effects associated with the use of Rilpivirine are-
- Depressive disorders
- Headache
- Insomnia
- Rashes
- Abdominal pain
- Drowsiness
- Vomiting
- Fatigue
- Dizziness
Some serious side effects include:
- rash along with fever, swelling of the face, lips, mouth, tongue, or throat, blisters, trouble breathing or swallowing, mouth sores, redness or swelling of the eyes (conjunctivitis), pain on the right side of the stomach (abdominal) area, or dark colored urine
FAQs
Q. What kind of drug is rilpivirine?
Rilpivirine is in a group of medications called non-nucleoside reverse transcriptase inhibitors (NNRTIs). It works by reducing the amount of HIV in the blood.
Q. When was rilpivirine FDA approved?
On May 20, 2011, the US Food and Drug Administration (FDA) approved EDURANT (rilpivirine) tablets for use with other antiretroviral agents (ARVs) to treat human immunodeficiency virus type 1 (HIV-1) in adults who have never had HIV therapy (treatment-naive).
